Iron
General Properties, Sources Of Iron, How Iron Is Obtained, How We Use Iron, Biochemical Applications
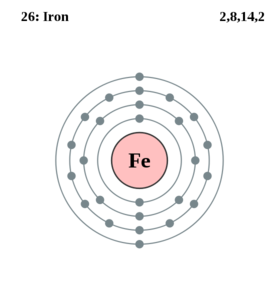
Iron is a metallic chemical element of atomic number 26. Its symbol is Fe, atomic weight is 55.847, specific gravity is 7.874, melting point is 2,795°F (1,535°C), and boiling point is 4,982°F (2,750°C).
Iron is one of the transition metals, occurring in group 8 of the periodic table. Four naturally occurring isotopes exist with atomic weights of 54 (5.8%), 56 (91.7%), 57 (2.2%), and 58 (0.3%). In addition, six radioactive isotopes have been prepared, with atomic weights of 52, 53, 55, 59, 60, and 61. The element was originally known by its Latin name ferrum, from which its chemical symbol is derived.
Additional topics
- Iron - General Properties
- Iron - Sources Of Iron
- Iron - How Iron Is Obtained
- Iron - How We Use Iron
- Iron - Biochemical Applications
- Iron - Chemistry And Compounds
- Other Free Encyclopedias
Science EncyclopediaScience & Philosophy: Intuitionist logic to Kabbalah