Boiling Point
The boiling point of a liquid substance is the temperature at which the vapor pressure of the liquid equals the external pressure on the liquid. Vapor bubbles form in the liquid, rise to the surface and burst, causing the liquid to boil.
At room temperature, in a closed system, there is an equilibrium between the liquid and its vapor phase. For example, if a glass of water is left open, the water will eventually evaporate, although it may take a few days. On the other hand, if the glass is kept covered, when the cover is removed, there is a large amount of water on the bottom of the cover. The molecules of the liquid in the glass move into the vapor phase, but when they encounter the cold surface of the cover, they condense back into liquid form and fall back into the liquid. When the rate of movement of molecules into the gas phase from the liquid equals the rate of movement of molecules into the liquid phase from the gas phase, the two phases are in equilibrium.
When the temperature is raised, more molecules have enough energy to enter the gas phase. If the glass were left uncovered, the water would disappear more quickly. If the glass was kept covered, a new equilibrium would be established more quickly. As the temperature increases, more molecules enter the gas phase and the vapor pressure of the liquid increases.
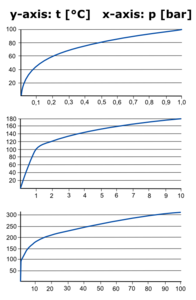
At some point in time, as the temperature increases, the vapor pressure of the liquid will increase to the point where it equals the external or atmospheric pressure. At this point, bubbles begin to form and rise in the liquid and it is said to be boiling. Before this temperature, bubbles cannot form because the external pressure is greater than the pressure in the bubble and it collapses. The temperature at which the external pressure and the vapor pressure are equal is called the normal boiling point. For water, this temperature is 212°F (100°C). In general, a liquid with high vapor pressures and a low normal boiling point is said to be volatile. Such liquids usually have strong smells. Liquids with low vapor pressures and high normal boiling points are non-volatile and have little or no odor.
If the external pressure is less than one atmosphere, liquids will boil at lower temperatures than their normal boiling points. At high elevations, the atmospheric pressure is much lower than one atmosphere. At the top of Mount Everest, where the atmospheric pressure is about 5 psi (260 mm Hg), the boiling point of water is only 160°F (71°C). At such high elevations, it is often necessary to follow special instructions for cooking and baking, as the water temperature is not high enough to cook food. Conversely, if the atmospheric pressure is greater than one atmosphere, liquids will boil at higher temperatures than their normal boiling points. We can use this to our advantage. In a pressure cooker, we increase the pressure so that it is greater than one atmosphere. As a result, water boils at a higher temperature and food cooks faster. We can also raise the boiling point of a liquid by adding a non-volatile solute to it. For example, adding salt to water raises its boiling point.
Rashmi Venkateswaran
Additional topics
Science EncyclopediaScience & Philosophy: Bilateral symmetry to Boolean algebra