Cellular Respiration
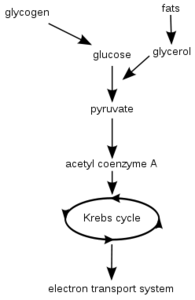
Cellular respiration in the presence of oxygen (aerobic respiration) is the process by which energy-rich organic substrates are broken down into carbon dioxide and water, with the release of a considerable amount of energy in the form of adenosine triphosphate (ATP). Anaerobic respiration breaks down glucose in the absence of oxygen, and produces pyruvate, which is then reduced to lactate or to ethanol and CO2. Anaerobic respiration releases only a small amount of energy (in the form of ATP) from the glucose molecule.
Respiration occurs in three stages. The first stage is glycolysis, which is a series of enzyme-controlled reactions that degrades glucose (a 6-carbon molecule) to pyruvate (a 3-carbon molecule) which is further oxidized to acetylcoenzyme A (acetyl CoA). Amino acids and fatty acids may also be oxidized to acetyl CoA as well as glucose.
In the second stage, acetyl CoA enters the citric acid (Krebs) cycle, where it is degraded to yield energy-rich hydrogen atoms which reduce the oxidized form of the coenzyme nicotinamide adenine dinucleotide (NAD+) to NADH, and reduce the coenzyme flavin adenine dinucleotide (FAD) to FADH2. (Reduction is the addition of electrons to a molecule, or the gain of hydrogen atoms, while oxidation is the loss of electrons or the addition of oxygen to a molecule.) Also in the second stage of cellular respiration, the carbon atoms of the intermediate metabolic products in the Krebs cycle are converted to carbon dioxide.
The third stage of cellular respiration occurs when the energy-rich hydrogen atoms are separated into protons [H+] and energy-rich electrons in the electron transport chain. At the beginning of the electron transport chain, the energy-rich hydrogen on NADH is removed from NADH, producing the oxidized coenzyme, NAD+ and a proton (H+) and two electrons (e-). The electrons are transferred along a chain of more than 15 different electron carrier molecules (known as the electron transport chain). These proteins are grouped into three large respiratory enzyme complexes, each of which contains proteins that span the mitochondrial membrane, securing the complexes into the inner membrane. Furthermore, each complex in the chain has a greater affinity for electrons than the complex before it. This increasing affinity drives the electrons down the chain until they are transferred all the way to the end where they meet the oxygen molecule, which has the greatest affinity of all for the electrons. The oxygen thus becomes reduced to H2O in the presence of hydrogen ions (protons), which were originally obtained from nutrient molecules through the process of oxidation.
During electron transport, much of the energy represented by the electrons is conserved during a process called oxidative phosphorylation. This process uses the energy of the electrons to phosphorylate (add a phosphate group) adenosine diphosphate (ADP), to form the energy-rich molecule ATP.
Oxidative phosphorylation is driven by the energy released by the electrons as they pass from the hydrogens of the coenzymes down the respiratory chain in the inner membrane of the mitochondrion. This energy is used to pump protons (H+) across the inner membrane from the matrix to the intermediate space. This sets up a concentration gradient along which substances flow from high to low concentration, while a simultaneous current of OH- flows across the membrane in the opposite direction. The simultaneous opposite flow of positive and negative ions across the mitochondrial membrane sets up an electrochemical proton gradient. The flow of protons down this gradient drives a membrane-bound enzyme, ATP synthetase, which catalyzes the phosphorylation of ADP to ATP.
This highly efficient, energy conserving series of reactions would not be possible in eukaryotic cells without the organelles called mitochondria. Mitochondria are the "powerhouses" of the eukaryotic cells, and are bounded by two membranes, which create two separate compartments: an internal space and a narrow intermembrane space. The enzymes of the matrix include those that catalyze the conversion of pyruvate and fatty acids to acetyl CoA, as well as the enzymes of the Krebs cycle. The enzymes of the respiratory chain are embedded in the inner mitochondrial membrane, which is the site of oxidative phosphorylation and the production of ATP.
In the absence of mitochondria, animal cells would be limited to glycolysis for their energy needs, which releases only a small fraction of the energy potentially available from the glucose.
The reactions of glycolysis require the input of two ATP molecules and produce four ATP molecules for a net gain of only two molecules per molecule of glucose. These ATP molecules are formed when phosphate groups are removed from phosphorylated intermediate products of glycolysis and transferred to ADP, a process called substrate level phosphorylation (synthesis of ATP by direct transfer of a high-energy phosphate group from a molecule in a metabolic pathway to ADP).
In contrast, mitochondria supplied with oxygen produce about 36 molecules of ATP for each molecule of glucose oxidized. Procaryotic cells, such a bacteria, lack mitochondria as well as nuclear membranes. Fatty acids and amino acids when transported into the mitochondria are degraded into the two-carbon acetyl group on acetyl CoA, which then enters the Krebs cycle. In animals, the body stores fattyacids in the form of fats, and glucose in the form of glycogen in order to ensure a steady supply of these nutrients for respiration.
While the Krebs cycle is an integral part of aerobic metabolism, the production of NADH and FADH 2 is not dependent on oxygen. Rather, oxygen is used at the end of the electron transport chain to combine with electrons removed from NADH and FADH2 and with hydrogen ions in the cytosol to produce water.
Although the production of water is necessary to keep the process of electron transport chain in motion, the energy used to make ATP is derived from a different process called chemiosmosis.
Chemiosmosis is a mechanism that uses the proton gradient across the membrane to generate ATP and is initiated by the activity of the electron transport chain. Chemiosmosis represents a link between the chemical and osmotic processes in the mitochondrion that occur during respiration.
The electrons that are transported down the respiratory chain on the mitochondrion's inner membrane release energy that is used to pump protons (H+) across the inner membrane from the mitochondrial matrix into the intermembrane space. The resulting gradient of protons across the mitochondrial inner membrane creates a backflow of protons back across the membrane. This flow of electrons across the membrane, like a waterfall used to power an electric turbine, drives a membrane-bound enzyme, ATP synthetase. This enzyme catalyzes the phosphorylation of ADP to ATP, which completes the part of cellular respiration called oxidative phosphorylation. The protons, in turn, neutralize the negative charges created by the addition of electrons to oxygen molecules, with the resultant production of water.
Cellular respiration produces three molecules of ATP per pair of electrons in NADH, while the pair of electrons in FADH2 generate two molecules of ATP. This means that 12 molecules of ATP are formed for each acetyl CoA molecule that enters the Krebs cycle; and since two acetyl CoA molecules are formed from each molecule of glucose, a total of 24 molecules of ATP are produced from each molecule of this sugar. When added to the energy conserved from the reactions occurring before acetyl CoA is formed, the complete oxidation of a glucose molecule gives a net yield of about 36 ATP molecules. When fats are burned, instead of glucose, the total yield from one molecule of palmitate, a 16-carbon fatty, is 129 ATP.
See also Catabolism; Respiration.
Resources
Books
Alberts, Bruce, et al. Molecular Biology of The Cell. 2nd ed. New York: Garland Publishing, 1989.
Lehninger, Albert L. Principles of Biochemistry. New York: Worth Publishers, 1982.
Marc Kusinitz
Additional topics
Science EncyclopediaScience & Philosophy: Categorical judgement to Chimaera